 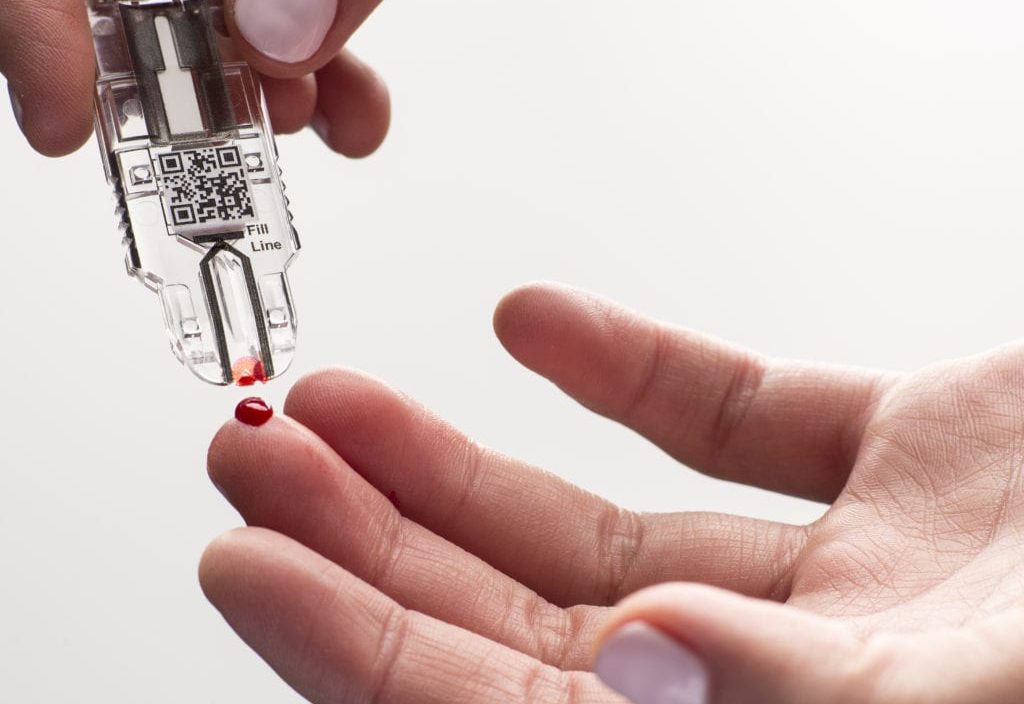
In addition, many firms are concentrating on creating POC platforms that rely on nanoparticles. One of the main things pushing point-of-care testing forward, especially for infectious disease diagnosis, is lab-on-a-chip technology. Innovations have facilitated the development of new techniques for these types of diagnostics in the miniaturization of chip technology, microfluidics, and biosensors. The groundwork for future revolutions has been laid by cloud-based deep learning systems. Lab-on-a-chip platforms, wearable technology, and smartphone-based technology innovations have contributed significantly to the development of POCT devices in recent years. Major market players have shifted their attention to adopting cutting-edge technologies like artificial intelligence to create effective point-of-care diagnostics. What are the Future Opportunities for Point-Of-Care Molecular Diagnostics? Technological Advancement As a result, during the predicted period, the global market is expected to grow slower than usual due to the lack of accuracy in POC testing. It may lead to incorrect treatment of hypoglycaemia or a delay in diagnosing hyperglycaemia. This device is used to quantitatively test whole blood that has not been pre-treated with anticoagulants in the laboratory or at the point of care. For instance, Siemens Healthcare issued a Class 2 Device Recall for the BGEM Test Card In-vitro diagnostic device in August 2020. Pre-analytical errors plague the field of point-of-care testing. The high price tag of virus-clearing technologies has limited the worldwide lack of accuracy of rapid point-of-care diagnostic tests, lowering the number of FDA approvals for rapid tests and thereby slowing the expansion of the market. What are the Main Factors that will slow down the Market for Point-Of-Care Molecular Diagnostics? Lack of Accuracy of POC Diagnostics Therefore, the need for a decentralized healthcare setting has been growing in response to the rising demand for quick and accurate diagnostics performed at home or in the doctor's office for patient convenience. A proliferation of companies catering to the rapid turnaround testing market has developed infectious disease tests for remote laboratories. Decentralizing testing for chronic and infectious diseases has given patients more accessible access to diagnostics, which had previously only been available at a single point of care. Point-of-Care testing has emerged as a vital component of patient-centered healthcare due to the urgent need for timely diagnosis results that underpin effective and convenient treatment planning. 
Proliferation of Decentralized Healthcare Systems The rising incidence of diabetes and other chronic diseases worldwide is expected to boost the point-of-care diagnostics market as more people seek effective means of diagnosing and treating these conditions. Lower respiratory tract infections caused by the respiratory syncytial virus (RSV) affect between 4 and 5 million children annually, according to the Centers for Disease Control and Prevention (CDC). One of the main factors driving demand for point-of-care diagnostics during the projected timeframe is the high prevalence rate of such conditions. There has been a worldwide rise in the prevalence of chronic diseases like cardiovascular disorders, diabetes, and various infectious diseases. Market Dynamics What are the Primary Factors driving the Market for Point-Of-Care Molecular Diagnostics? Growing Incidence of Chronic and Infectious Diseases 
The healthcare industry relies on it for disease detection and urgent use authorization. The detection of specific sequences of DNA or RNA in microorganisms is the basis of this molecular diagnostic. Several factors, including increased consumer awareness of molecular mechanisms, rare disorders, and technological advances in DNA sequencing, are propelling the market for point-of-care molecular diagnostics. However, the limitations of molecular diagnostics have prompted them to seek out alternative methods of diagnosis due to issues like the tests' high costs, their time-consuming nature, and the delays in their results. The field of molecular diagnostics has been widely utilized by doctors in hospitals, clinics, and other medical facilities for quite some time. Physicians can raise the quality of care they provide with the help of POC molecular diagnostic testing by quickly diagnosing and treating patients. It is projected to reach USD 4487.93 million by 2031, growing at a CAGR of 7.84% during the forecast period (2023–2031). The global point-of-care molecular diagnostics market was valued at USD 2275.24 million in 2022. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
